While microalgae may be a new term for some people, everyone will have seen them. When a green biomass grows on a pond (sometimes called pondscum!) that’s microalgae. Microalgae, including cyanobacteria and diatoms, are mostly single-celled microorganisms, which only need sunlight, water and a small amount of inorganic nutrients to make its own food, similar to plants. Microalgae are at the base of the aquatic food web and can contribute up to 40% of global productivity. They have a much higher growth rate compared to land plants but don’t require arable land or depend as much on weather conditions.
Microalgae can be mass-cultivated for a wide range of applications (Figure 1). They have the potential to satisfy many global demands; however, delivering this potential requires improvements in cultivation techniques.
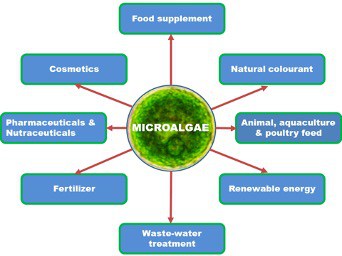
- Food Supplement: Spirulina and Chlorella are the most commercially used species in human nutrition due to their high protein content and excellent nutritive values. Spirulina is a source of high-quality protein, γ-linolenic acid, and pigments; Chlorella is composed of a variety of substances, including nucleic acids, amino acids, vitamins, minerals, polysaccharides, glycoproteins, and β-glucans with potential antioxidant, anti-diabetic, anti-cancer, anti-viral and antibacterial activities.
- Natural Colourant for use in food, cosmetics, and pharmaceuticals as substitutes for synthetic dyes. Some commonly used synthetic colours are allergens, irritants or may be carcinogenic. Natural colorants from microalgae can be used as alternatives to these. Major pigments of the microalgae which are used as colourants are carotenoids and phycobiliproteins.
- Animal, aquaculture and poultry feed to improve animaland aquaculture growth, fish colour and meat quality. 30% of microalgae sold is for animal feed applications and over 50% of the current world production of Spirulina is used as a feed supplement. Research has shown that microalgae have a positive effect on the physiology and the external appearance of animals. Microalgae-based feed can improve the quality of aquaculture, poultry and animal growth, and thus productivity.
- Fertilizer as a promising alternative to chemical and synthetic fertilizers. Nearly all cyanobacteria can be used as biofertilizers to increase crop production as they are sources of the main macro minerals such as nitrogen and phosphate along with essential micro-minerals.
- Waste-water treatment by removal of nutrients such as nitrogen and phosphorus to prevent eutrophication and water pollution. The ability of microalgae to grow in wastewater is an economic, cost-effective, and eco-friendly treatment process compared to conventional treatment methods. Conventional methods are dependent on high energy costs and treatment chemicals, whereas with microalgal treatment, pollutants can be converted to a valuable biomass while fixing atmospheric CO
- Renewable energy for environmentally friendly energy resources such as biodiesel, bioethanol and biogas. Compared with other renewable energy sources such as wind, geothermal energy, solar energy or hydropower, microalgal biomass is a more controlled and stable source for production of energy. Moreover, compared to other biomass sources, their oil content and yield are 15-300 times greater than traditional crops. Microalgae have higher growth rates than terrestrial plants and can be cultivated on non-arable lands in freshwater, seawater, and wastewater.
- Pharmaceuticals and Nutraceuticals due to their high bioactive properties they have the potential for the development of pharmaceuticals and nutraceuticals. These include β-carotene, astaxanthin, fucoxanthin, docosahexaenoic acid (DHA), eicosapentaenoic acid (EPA), γ-linolenic acid, C-phycocyanin, and bioactive and functional molecules, which have been reported to possess anticancer, antibacterial, and antiviral properties. Microalgae that have been cultivated commercially for this sector are Spirulina, Dunaliella sp., Chlorella sp., and Haematococcus pluvialis.
- Cosmetics due to their bioactive content. Microalgal products have been used in the cosmetic industry as antioxidants, sunscreens and moisturizing agents. Microalgae contian a diverse range of bioactive compounds such as proteins, lipids, vitamins (A, B1, B2, B6, B12, C), pigments (β-carotene, astaxanthin, lutein), phycobiliproteins (phycocyanin, phycoerythrin) and mycosporine-like amino acids (MAAs).
R&D facilities at Shannon ABC
Most microalgae are photoautotrophs, requiring mainly sun or artificial light, water, and inorganic nutrients for growth, and can be produced rapidly on non–arable land such as closed or open bioreactors in lab-scale and/or large-scale volumes. For microalgae cultivation, Shannon ABC has different growth chambers in operation at various culture volumes.
1) Environmental Growth Chambers
Environmental growth chambers (EGC) provide a controlled environment to analyse the effects of environmental parameters to optimise microalgal growth and physiology. These include:
- Light intensity
- Light/dark cycle
- Temperature
- Humidity
- Air
- Carbon dioxide.
Once the ideal environmental parameters for growth are identified, the productivity of the biomass can be optimised. The walk in EGC in Shannon ABC is 4.5m2 and allows walk-in activities and multiple R&D experimental set-ups in a single chamber at any given time (Figures 2&3).


2) Reach-in Growth Chamber
In the same way as the EGC, the reach-in growth chamber also provides homogeneous light, temperature and humidity distribution within the chamber (Figure 4). The environmental parameters can be controlled to achieve ideal growth conditions for the microalgae and cyanobacteria to deliver enhanced biomass and bioactive ingredients. The reach-in chamber is mostly for small-scale cultivation and maintenance of seed cultures required for various R&D experiments.


3) 100 L Photobioreactors with LED lighting
This custom-designed 100L photobioreactor (Figure 5) is used for the optimisation of pilot-scale cultivation of cyanobacteria/microalgae for specific target molecules, for both food and cosmetics applications. It uses LED lighting technology for the enhancement of microalgal pigment production.

Microalgal biomass harvesting:
After the desired growth is achieved, microalgae need to be separated from the growth media for further applications. Flocculation, centrifugation and filtration are the most suitable harvesting techniques depending on the type of microalgae (Figure 6). Flocculation is a physical method for concentrating the microalgal biomass by bringing small particles into bigger flocs for a quicker sedimentation, before further dewatering processes such as centrifugation and filtration. Centrifugation is the most commonly used technique to separate algal biomass suspended in a liquid medium and is more suited for small-volumes. Finally, vacuum filtration is a fast filtration technique where air is forced through the filter paper by the application of reduced pressure. The biomass can then be removed from the filter paper.

Biomass extraction and biochemical analysis:
Shannon ABC has a dedicated suite for extraction of biomass and biochemical and bioanalytical analysis. After the algal biomass is harvested, the desired compounds are extracted using water, organic solvent or by using supercritical CO2 fluid extraction (SCFE), a green extraction technology. Lipid soluble compounds such as carotenoids (astaxanthin, β-carotene, lutein, zeaxathin, fucoxanthin, canthaxanthin), lipids and fatty acids (omega-3 and omega-6), polyphenols etc. can be extracted by SCFE (Figure 7).

Instruments such as LC-MS, GC-MS and HPLC are used for the analysis of carotenoids, fatty acids (EPA, DHA, palmitic acid, linolenic acid, etc), sugars (mono-, di-, poly-saccharides, and FOS), amino acids (GABA, ergothioneine, MAAs, etc.), polyphenols, etc. The biochemical and bioactivity assays for microalgal biomass and their active extracts include the estimation of protein, carbohydrate, lipids, and pigment content, antioxidants activities (DPPH, FRAP, ORAC) and various other enzyme activities.
Eventually, the biomass feedstock (Figure 8) and extracts can be used for various applications such as yoghurt, cheese, lip balm, soap, and cream (Figure 9).

Figure 8. Microalgal fresh biomass after harvesting after dewatering and centrifugation

Figure 9. Application studies carried out at Shannon ABC using microalgal ingredients.
In conclusion, microalgae have a wide range of biotechnological applications, including animal and human nutrition, biofertilizer, wastewater treatment, biofuels and bioactive compounds. Thus, microalgae and their high-value compounds have great potential for interested biotechnological companies who prefer sustainable natural resources and innovation in their product range.
If you would like more information on microalgal biotechnology and R&D that may be relevant for your company, please contact Dr Sushanta Kumar Saha, Senior Microalgal Biotechnologist (Sushanta.Saha@lit.ie), or Dr Patrick Murray, Head of Research and Technology Transfer at Limerick Institute of Technology (patrick.murray@lit.ie) or Hande Ermis, Research Assistant (hande.ermis@lit.ie).



